Mining in Manitoba
Methane Hydrates
FUTURE METHODS
Ocean Mining
Deep Sea Nodules
Biological Leaching
Asteroid Mining
Helium-3 Mining
Nuclear Blasting
Moon Mining
Oxygen Mining
Gold from the Sea
Methane hydrates are a type of natural formation that contains
large amounts of methane, which is also known as natural gas, and water, in the form of
ice. Hydrates are plentiful in nature, both underwater and under permafrost. They are a
potential source - possibly a very important source -- of energy for the future. However,
little is currently known about cost-effective ways to turn hydrates into an energy
resource.

From a scientist's point of view, methane hydrates are cages of water molecules that surround and trap methane molecules. They are crystalline solids that form under moderate pressure (for instance, at water depths greater than 300 meters) and at temperatures that are low but above the freezing point of water. In nature, hydrate formations are very hard and ice-like, and they may contain molecules of substances other than methane. When hydrates are made in the laboratory or classroom, they tend to look more like slush.
- Hydrates have potential as a future energy resource
- Hydrates may be a source and also a sink for atmospheric methane Better understanding of these natural deposits may increase our understanding of climate change.
- Hydrates affect the strength of the sediments in which they are found. Areas with hydrates appear to be less stable than other areas of the seafloor. Consequently, it is important to assess their presence prior to the construction of underwater structures related to military defense and to gas and oil exploration and production. Lack of stability might also be a factor in climate change
Where do methane hydrates form?
Stable methane hydrates are found at the temperature and pressure conditions that exist near and just beneath the sea floor where water depths exceed 300 to 500 meters. Hydrate is also stable in conjunction with permafrost at high latitudes.
How are hydrates formed?
A. There are two processes:
- Most natural gas hydrate is formed from biogenic methane, excreted by bacteria that eat organic matter that has been washed into (or died in) the ocean. This type of hydrate is concentrated where there is a rapid accumulation of organic detritus and also where there is a rapid accumulation of sediments (which protect detritus from oxidation).

- Hydrates also form when faults permit natural gas (or other gases) to migrate from deeper inside the Earth's crust to the surface of the seabed at places with appropriate temperature and pressure levels.
These processes can also cause hydrates to form below permafrost, which acts as a cap to prevent further upward migration of gas into the atmosphere.

Scientists generally believe that most natural gas hydrate is formed from biogenic methane (produced by bacteria), and that therefore it is concentrated 1) where there is a rapid accumulation of dead organic material (from which bacteria generate methane) and 2) where there is a rapid accumulation of sediments, which protect the material from oxidation.
Geologists tend to believe that deposits formed through underground venting will prove the easiest to recover because there would seem to be more potential for biologically produced hydrates to be distributed relatively evenly over a wide area, while a vent is localized. Additionally, there appears to be more potential that free methane is trapped underneath hydrates formed at vents.
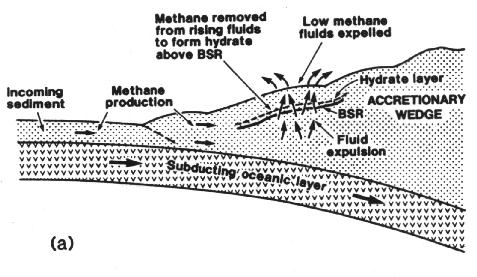
This speculation makes sense given the characteristics of current gas and oil production. Methane, for instance, one of the most common of earth's molecules, is produced naturally by bacterial action in swamps, landfills, rice paddies, and the digestive tracts of cows and termites. Generally, however, the concentration of methane in any one place is so low that - with the exception of a few landfills - we do not attempt to capture this "resource." Similarly, oil is frequently dispersed throughout shale formations; but processing the shale is an energy-intensive activity in itself, thus raising production costs far above those involving more porous formations.